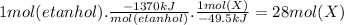Physics, 11.11.2019 22:31 adiboo2004
Assume that the complete combustion of one mole of ethanol to carbon dioxide and water liberates 1370 kj of energy (δ°′=−1370 kj/mol ). if the energy generated by the combustion of ethanol is entirely converted to the synthesis of a hypothetical compound x, calculate the number of moles of the compound that could theoretically be generated. use the value δ°′compound x=−45.9 kj/mol . round your answer to two significant figures.

Answers: 1


Another question on Physics


Physics, 22.06.2019 11:30
What is the name for the remnant of an asymptotic giant that has lost its shells? black dwarf white dwarf yellow giant black hole
Answers: 3

Physics, 22.06.2019 19:30
A47.2 g block of copper whose temperature is 480 k is placed in an insulating box with a 91.8 g block of lead whose temperature is 200 k. (a) what is the equilibrium temperature of the two-block system? (b) what is the change in the internal energy of the two-block system between the initial state and the equilibrium state? (c) what is the change in the entropy of the two-block system? the heat capacities of copper and lead are 386 j/kg·k and 128 j/kg·k, respectively.
Answers: 1

Physics, 22.06.2019 20:30
Aball is thrown from the top of a building with an initial velocity of 21.9 m/s straight upward, at an initial height of 51.6 m above the ground. the ball just misses the edge of the roof on its way down, as shown in the figure. (a) determine the time needed for the ball to reach its maximum height. (b) determine the maximum height. (c) determine the time needed for the ball to return to the height from which it was thrown, and the velocity of the ball at that instant. (d) determine the time needed for the ball to reach the ground. (e) determine the velocity and position of the ball at t = 5.35 s.
Answers: 1
You know the right answer?
Assume that the complete combustion of one mole of ethanol to carbon dioxide and water liberates 137...
Questions







Mathematics, 14.04.2020 21:46



Social Studies, 14.04.2020 21:46





Mathematics, 14.04.2020 21:46





Mathematics, 14.04.2020 21:46