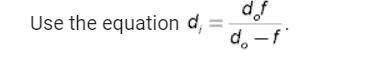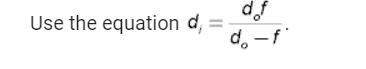Answers: 1


Another question on Physics

Physics, 22.06.2019 05:50
Acylinder with a movable piston contains 11.7 moles of a monatomic ideal gas at a pressure of 1.32×10^5 pa. the gas is initially at a temperature of 300 k. an electric heater adds 43200 j of energy into the gas while the piston moves in such a way that the pressure remains constant. cp=20.79 j k^−1 mol^−1 for a monatomic ideal gas, and that the number of gas molecules is equal to avogadro's number (6.022×10^23) times the number of moles of the gas. (a) what is the temperature of the gas after the energy is added? (b) what is the change in volume of the gas? (c) how much work is done by the gas during this process?
Answers: 3

Physics, 22.06.2019 09:40
(a) assume the equation x = at^3 + bt describes the motion of a particular object, with x having the dimension of length and t having the dimension of time. determine the dimensions of the constants a and b. (use the following as necessary: l and t, where l is the unit of length and t is the unit of time.) (b) determine the dimensions of the derivative dx/dt = 3at^2 + b. (use the following as necessary: l and t, where l is the unit of length and t is the unit of time.)
Answers: 1


Physics, 22.06.2019 16:30
When conducting an investigation to determine which type of fertilizer produces the most flowers on a plant, which variable would be graphed on the x-axis once you collected the data? a. type of fertilizer b. amount of flowers c. amount of water d. number of days that the plants grew
Answers: 1
You know the right answer?
An object is located 5.0 cm from a convex lens. the lens focuses light at a distance of 10.0 cm. wha...
Questions

History, 19.10.2019 07:30



Social Studies, 19.10.2019 07:30


History, 19.10.2019 07:30


Geography, 19.10.2019 07:30


Biology, 19.10.2019 07:30

Mathematics, 19.10.2019 07:30





Mathematics, 19.10.2019 07:30


History, 19.10.2019 07:30

Business, 19.10.2019 07:30

English, 19.10.2019 07:30