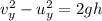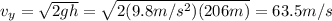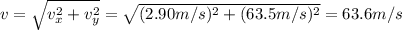Answers: 2


Another question on Physics

Physics, 22.06.2019 03:30
As part of an industrial process, air as an ideal gas at 10 bar, 400k expands at steady state through a valve to a pressure of 4 bar. the mass flow rate of air is 0.5 kg/s. the air then passes through a heat exchanger where it is cooled to a temperature of 295k with negligible change in pressure. the valve can be modeled as a throttling process, and kinetic and potential energy effects can be neglected. (a) for a control volume enclosing the valve and heat exchanger and enough of the local surroundings that the heat transfer occurs at the ambient temperature of 295 k, determine the rate of entropy production, in kw/k. (b) if the expansion valve were replaced by an adiabatic turbine operating isentropically, what would be the entropy production? compare the results of parts (a) and (b) and discuss.
Answers: 3


Physics, 22.06.2019 17:30
How does the entropy of steam compare to the entropy of ice?
Answers: 2

Physics, 22.06.2019 22:00
Should we celebrate the voyages of zheng he mini q answer key
Answers: 3
You know the right answer?
Multnomah falls in oregon are approximately 206m high. if the columbia river is flowing horizontally...
Questions



Chemistry, 20.01.2020 09:31


Mathematics, 20.01.2020 09:31

English, 20.01.2020 09:31

Physics, 20.01.2020 09:31

History, 20.01.2020 09:31


Social Studies, 20.01.2020 09:31

History, 20.01.2020 09:31




Social Studies, 20.01.2020 09:31


Mathematics, 20.01.2020 09:31



Mathematics, 20.01.2020 09:31

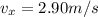 , since there are no forces acting along the horizontal direction
, since there are no forces acting along the horizontal direction