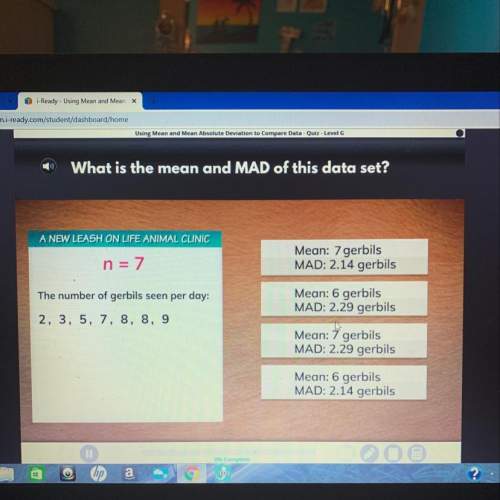
Mathematics, 02.12.2021 01:00 mtassy85
The pH of a solution is a measure of its acidity or alkalinity. Specifically, pH = -log[H+] where [H+] is the hydrogen ion concentration in moles per liter. A solution with a pH less than 7 is an acid, one with a pH greater than 7 is a base (alkaline), and a pH of 7 is regarded as neutral.
a)The hydrogen ion concentration of pure water is [H+] = 10-7 moles per liter. Find its pH.
b) Find the pH of a solution with [H+] = 6.3 × 10-13 moles per liter.
c) The pH of gastric acid (the acid in your stomach) is about 0.7. What is the corresponding hydrogen ion concentration?

Answers: 3


Another question on Mathematics

Mathematics, 21.06.2019 13:30
Based upon past experience, barry expects no overdrafts. he expects no 2nd copies of statements. barry estimates that he will use network atms about 5 times a month with either bank. barry decides in the end to choose eecu. assuming that both banks provide the necessary services equally well, and based upon the tables of fees given above, how much can barry reasonably expect to save annually by choosing eecu in this case over e-town bank? a. $72 b. $78 c. $144 d. $24
Answers: 2

Mathematics, 21.06.2019 16:00
Does the problem involve permutations or? combinations? do not solve. the matching section of an exam has 4 questions and 7 possible answers. in how many different ways can a student answer the 4 ? questions, if none of the answer choices can be? repeated?
Answers: 1


Mathematics, 21.06.2019 17:00
Describe what moves you could use to create the transformation of the original image shown at right
Answers: 1
You know the right answer?
The pH of a solution is a measure of its acidity or alkalinity. Specifically, pH = -log[H+] where [H...
Questions

Mathematics, 10.06.2021 16:00


Mathematics, 10.06.2021 16:00


Law, 10.06.2021 16:00

Social Studies, 10.06.2021 16:00


Mathematics, 10.06.2021 16:00

Social Studies, 10.06.2021 16:00


Mathematics, 10.06.2021 16:00

Biology, 10.06.2021 16:00


Chemistry, 10.06.2021 16:00

Mathematics, 10.06.2021 16:00



Mathematics, 10.06.2021 16:00