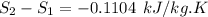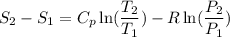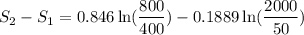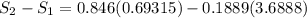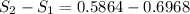Engineering, 14.07.2020 14:01 sandram74691
Carbon dioxide initially at 50 kPa, 400 K, undergoes a process in a closed system until its pressure and temperature are 2 MPa and 800 K, respectively. Assuming an ideal gas behaviour, find the entropy change of the carbon dioxide by assuming that the specific heats are constant. For the gas, take Cp = 0.846 kJ/kg. K and R = 0.1889 kJ/kg. K

Answers: 3


Another question on Engineering

Engineering, 04.07.2019 18:10
Carbon dioxide gas expands isotherm a turbine from 1 mpa, 500 k at 200 kpa. assuming the ideal gas model and neglecting the kinetic and potential energies, determine the change in entropy, heat transfer and work for each kilogram of co2.
Answers: 2

Engineering, 04.07.2019 18:10
A-mn has a cubic structure with a0 0.8931 nm and a density of 7.47 g/cm3. b-mn has a different cubic structure, with a0 0.6326 nm and a density of 7.26 g/cm3. the atomic weight of manganese is 54.938 g/mol and the atomic radius is 0.112 nm. determine the percent volume change that would occur if a-mn transforms to b-mn.
Answers: 2

Engineering, 04.07.2019 18:10
Which from the following instrument is commonly used to detect the high pitch butzing sound in bearings? [clo4] a)-digital ultrasonic meter b)-infrared camera c)-spectroscopic d)-vibrometer
Answers: 2

Engineering, 04.07.2019 18:20
Inadequate stores control is not an obstacle to effective work order system. (clo4) a)-true b)-false
Answers: 3
You know the right answer?
Carbon dioxide initially at 50 kPa, 400 K, undergoes a process in a closed system until its pressure...
Questions







English, 23.07.2019 04:00

English, 23.07.2019 04:00




English, 23.07.2019 04:00




History, 23.07.2019 04:00


Chemistry, 23.07.2019 04:00