
Engineering, 29.09.2019 02:30 daii128
Consider the expansion of a gas at a constant temperature in a water-cooled piston-cylinder system. the constant temperature is achieved by controlled input of energy as heat q to the gas. treating the gas as idea, derive expressions for the energy output as work, w and energy input as heat, q, as a function of the expansion ratio. (v2/v1)

Answers: 2


Another question on Engineering

Engineering, 03.07.2019 15:10
Two flowing streams of argon gas are adiabatically mixed to form a single flow/stream. one stream is 1.5 kg/s at 400 kpa and 200 c while the second stream is 2kg/s at 500 kpa and 100 ? . it is stated that the exit state of the mixed single flow of argon gas is 150 c and 300 kpa. assuming there is no work output or input during the mixing process, does this process violate either the first or the second law or both? explain and state all your assumptions.
Answers: 1

Engineering, 04.07.2019 18:10
The thermal expansion or contraction of a given metal is a function of the f a)-density b)-initial temperature c)- temperature difference d)- linear coefficient of thermal expansion e)- final temperature f)- original length
Answers: 2

Engineering, 04.07.2019 18:10
Awall of 0.5m thickness is to be constructed from a material which has average thermal conductivity of 1.4 w/mk. the wall is to be insulated with a material having an average thermal conductivity of 0.35 w/mk so that heat loss per square meter shall not exceed 1450 w. assume inner wall surface temperature of 1200°c and outer surface temperature of the insulation to be 15°c. calculate the thickness of insulation required.
Answers: 3

Engineering, 04.07.2019 18:20
Determine the damped natural frequencies and the steady state response of a decoupled damped forced two degrees of freedom system. 10ä1 + 2q1 20q1 10 cos t; 10q2 +4q2 + 40q2 10 cos t
Answers: 3
You know the right answer?
Consider the expansion of a gas at a constant temperature in a water-cooled piston-cylinder system....
Questions




History, 18.10.2020 09:01

Social Studies, 18.10.2020 09:01

Mathematics, 18.10.2020 09:01

English, 18.10.2020 09:01




English, 18.10.2020 09:01

Chemistry, 18.10.2020 09:01

Chemistry, 18.10.2020 09:01


Mathematics, 18.10.2020 09:01



Mathematics, 18.10.2020 09:01


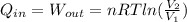
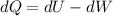
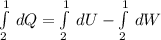
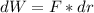
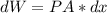
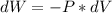
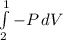
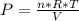 , so:
, so: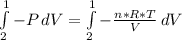
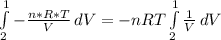
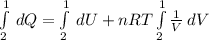
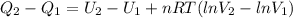
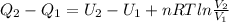
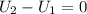 , so the heat exchanged to the system equals the work done by the system:
, so the heat exchanged to the system equals the work done by the system:

