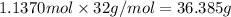Chemistry, 21.07.2019 08:00 rigobertogarza2
What mass of oxygen reacts when 84.9 g of iron is consumed in the following reaction: fe+o2= fe2o3

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:00
Calculate the mass of silver needed to react with chlorine to produce 126g if silver chloride?
Answers: 3

Chemistry, 22.06.2019 16:10
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1

Chemistry, 22.06.2019 19:30
Chlorine and water react to form hydrogen chloride and oxygen, like this: 2cl2 (g) + 2h2o (g) → 4hcl (g) + o2 (g) also, a chemist finds that at a certain temperature the equilibrium mixture of chlorine, water, hydrogen chloride, and oxygen has the following composition: compound concentration at equilibrium cl2 0.55m h2o 0.57m hcl 0.53m o2 0.34m calculate the value of the equilibrium constant kc for this reaction. round your answer to 2 significant digits.
Answers: 2

Chemistry, 22.06.2019 22:30
How do limiting factors most affect population size? ostop population growthrestrict population growthincrease population sizeresult in positive impactso
Answers: 1
You know the right answer?
What mass of oxygen reacts when 84.9 g of iron is consumed in the following reaction: fe+o2= fe2o3...
Questions

Mathematics, 11.07.2019 02:30


History, 11.07.2019 02:30

Health, 11.07.2019 02:30

Mathematics, 11.07.2019 02:30

Mathematics, 11.07.2019 02:30


Mathematics, 11.07.2019 02:30

Mathematics, 11.07.2019 02:30




Mathematics, 11.07.2019 02:30

Mathematics, 11.07.2019 02:30

Mathematics, 11.07.2019 02:30

Mathematics, 11.07.2019 02:30




English, 11.07.2019 02:30

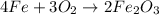
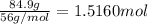
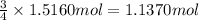 of oxygen gas
of oxygen gas