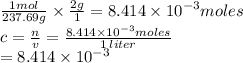Chemistry, 23.07.2019 06:00 helpmeplzandty
If you dissolve 2.00 g of nicl2*6h20 into a beaker of water and the final volume is 1.00 liters what will be the molar concentration of the solution?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 13:00
Using the thermodynamic information in the aleks data tab, calculate the standard reaction free energy of the following chemical reaction: →+p4o10s6h2ol4h3po4s round your answer to zero decimal places.
Answers: 3

Chemistry, 22.06.2019 19:30
Awoman's basketball has a circumference between 28.5 and 29.0 inches and a maximum weight of 20 ounces (two significant figures). what are these specifications in units of centimeters and grams?
Answers: 2

Chemistry, 22.06.2019 20:20
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3

Chemistry, 22.06.2019 21:20
If a simple machine aduces the strength of a force, what must be increased? the speed of the input force the work the simple machine performs the size of the simple machine the distance over which the force is applied
Answers: 1
You know the right answer?
If you dissolve 2.00 g of nicl2*6h20 into a beaker of water and the final volume is 1.00 liters what...
Questions


Mathematics, 02.03.2021 03:40


Mathematics, 02.03.2021 03:40

English, 02.03.2021 03:40




Chemistry, 02.03.2021 03:40

Mathematics, 02.03.2021 03:40

Mathematics, 02.03.2021 03:40


Social Studies, 02.03.2021 03:40




Mathematics, 02.03.2021 03:40