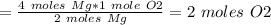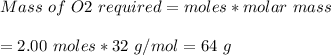Chemistry, 24.07.2019 03:30 carlinryan
The chemical equation below shows the burning of magnesium (mg) with oxygen (o2) to form magnesium oxide (mgo). 2mg + o2 2mgo the molar mass of o2 is 32.0 g/mol. what mass, in grams, of o2 is required to react completely with 4.00 mol of mg? a)2.00 b)64.0 c)128 d)256

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:30
Select the correct answer. when carbon dioxide dissolves in water, it sometimes reacts with water to form carbonic acid as in this balanced equation: co2 + h2o → h2co3. if 495 milliliters of carbon dioxide at 25°c and 101.3 kilopascals reacts with excess water, what is the theoretical yield of carbonic acid? use the periodic table and the ideal gas resource a. 0.889 g b. 1.10g c. 1.27g d. 2.02g what's the answer! quick!
Answers: 1

Chemistry, 22.06.2019 04:40
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3

Chemistry, 22.06.2019 11:40
Which of these expressions are correct variations of the combined gas law? p1v1t2 = p2v2t1 both
Answers: 2

Chemistry, 22.06.2019 19:30
If 16.00g of hydrogen gas reacts with 126.73g of oxygen, how many grams of water are yielded? (both reactants are completely consumed in the reaction.)
Answers: 2
You know the right answer?
The chemical equation below shows the burning of magnesium (mg) with oxygen (o2) to form magnesium o...
Questions

Mathematics, 13.11.2020 20:10


Chemistry, 13.11.2020 20:10



Mathematics, 13.11.2020 20:10






Biology, 13.11.2020 20:10

Biology, 13.11.2020 20:10

Arts, 13.11.2020 20:10

English, 13.11.2020 20:10



Mathematics, 13.11.2020 20:10

Biology, 13.11.2020 20:10

English, 13.11.2020 20:10