Chemistry, 24.07.2019 12:30 milkshakegrande101
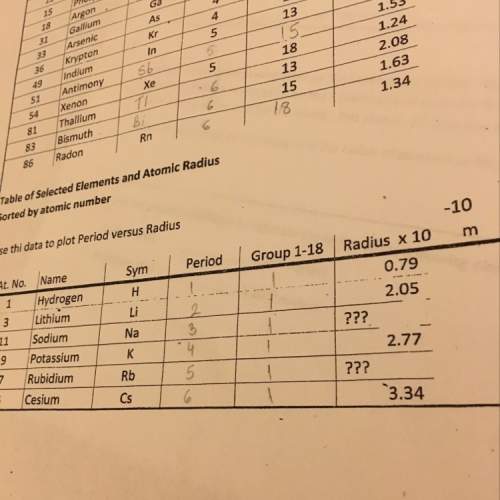
Pentaborane b5h9(s) burns vigorously in o2 to give b2o3(s) and h2o(l). what is δh° for the combustion of 1 mol of b5h9(s)? substance δh°f (kj/mol) b2o3(s) –1273.5 b5h9(s) +73.2 h2o(l) –285.8 a. -1486.1 kj b. -1632.5 kj c. -4396.7 kj d. -4652.85 kj e. -9086.1 kj

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:30
Why is soap used to remove grease? a. its nonpolar end dissolves the grease. b. it makes the water bond with the grease. c. it chemically bonds with the grease. d. its polar end dissolves the grease.correct answer for apex - a, its nonpolar end dissolves the grease.
Answers: 1

Chemistry, 22.06.2019 12:20
The yearly amounts of carbon emissions from cars in belgium are normally distributed with a mean of 13.9 gigagrams per year and a standard deviation of 5.8 gigagrams per year. find the probability that the amount of carbon emissions from cars in belgium for a randomly selected year are between 11.5 gigagrams and 14.0 gigagrams per year. a. 0.340 b. 0.660 c. 0.167 d. 0.397
Answers: 2

Chemistry, 22.06.2019 12:40
When 13.3 g koh is dissolved in 102.7 g of water in a coffee-cup calorimeter, the temperature rises from 21.4 °c to 31.53 °c. what is the enthalpy change per gram of koh (j/g) dissolved in the water? * take the density of water as 1.00 g/ml. * assume that the solution has a specific heat capacity of 4.18 j/g*k. enter to 1 decimal place. do not forget the appropriate sign /(+). canvas may auto-delete the (+) sign
Answers: 2

Chemistry, 22.06.2019 18:10
Measurements that have similar values are: a. usually accurate b. sometimes accurate c. always accurate d. never accurate
Answers: 1
You know the right answer?
Pentaborane b5h9(s) burns vigorously in o2 to give b2o3(s) and h2o(l). what is δh° for the combustio...
Questions

History, 29.10.2020 17:40


Biology, 29.10.2020 17:40

English, 29.10.2020 17:40

Mathematics, 29.10.2020 17:40


Social Studies, 29.10.2020 17:40


English, 29.10.2020 17:40





Mathematics, 29.10.2020 17:40

History, 29.10.2020 17:40


Mathematics, 29.10.2020 17:40

History, 29.10.2020 17:40

Mathematics, 29.10.2020 17:40

Chemistry, 29.10.2020 17:40

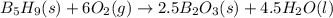
![\Delta H_r_x_n^0=[\sum \Delta H_f]_p_r_o_d_u_c_t-[\sum \Delta H_f]_r_e_a_c_t_a_n_t](/tpl/images/0127/2367/5178e.png)
![\Delta H_r_x_n^0=[2.5(-1273.5)+4.5(-285.8)]-[73.2+6(0)]](/tpl/images/0127/2367/81949.png)
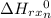 = -3183.75 - 1286.1 - 73.2
= -3183.75 - 1286.1 - 73.2