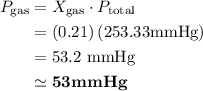Chemistry, 25.07.2019 07:00 ainhoagmz903
Atmospheric pressure at the summit of mount everest is about one third the pressure at sea level, which is 760 mm hg. if oxygen makes up 21% of the atmosphere by volume, the partial pressure of oxygen (po2) on mount everest is approximately atmospheric pressure at the summit of mount everest is about one third the pressure at sea level, which is 760 mm hg. if oxygen makes up 21% of the atmosphere by volume, the partial pressure of oxygen (po2) on mount everest is approximately 255 mm hg 53 mm hg 157 mm hg 760 mm hg

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 1

Chemistry, 22.06.2019 14:00
Ascientist measures the speed of sound in a monatomic gas to be 449 m/s at 20∘c. what is the molar mass of this gas?
Answers: 2

Chemistry, 22.06.2019 15:50
Elements in group 2 are all called alkaline earth metals. what is most similar about the alkaline earth metals?
Answers: 1

You know the right answer?
Atmospheric pressure at the summit of mount everest is about one third the pressure at sea level, wh...
Questions






History, 04.07.2019 11:00




History, 04.07.2019 11:00

English, 04.07.2019 11:00

History, 04.07.2019 11:00

History, 04.07.2019 11:00

Social Studies, 04.07.2019 11:00

Mathematics, 04.07.2019 11:00


History, 04.07.2019 11:00

Mathematics, 04.07.2019 11:00

History, 04.07.2019 11:00

History, 04.07.2019 11:00

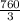 mmHg
mmHg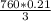 = 53.2 mmHg
= 53.2 mmHg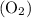 at Mount Everest is approximately 53 mmHg.
at Mount Everest is approximately 53 mmHg.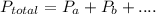 …… (1)
…… (1)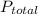 is total pressure of the gas mixture.
is total pressure of the gas mixture.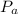 ,
, 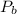 are the partial pressures of gas
are the partial pressures of gas  and
and 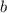 respectively.
respectively.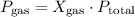 …… (2)
…… (2)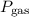 is the partial pressure of the gas.
is the partial pressure of the gas.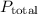 is the total pressure of the mixture.
is the total pressure of the mixture.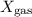 is the mole fraction of gas.
is the mole fraction of gas.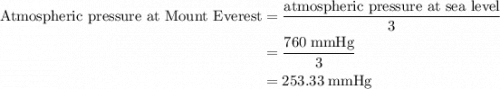
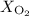 and 253.33 mmHg for
and 253.33 mmHg for 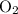 gas at Mount Everest.
gas at Mount Everest.