
Two moles of propene (c3h6) gas react with nine moles of oxygen (o2) gas to produce six moles of carbon dioxide gas (co2) and six moles of water gas (h2o). using this description of the reaction, write the corresponding chemical equation, showing the appropriate coefficients for each reactant and product. (include states-of-matter under the given conditions in your answer.)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Ibeg i need 20. a reaction produces 4.93 l of oxygen, but was supposed to produce 1 mol of oxygen. what is the percent yield?
Answers: 1

Chemistry, 22.06.2019 01:30
In a spacecraft, the following reaction occurs: co2(g) + 2lioh(s) -> lico3(s) + h2o(i) (i attached picture of equation) how many liters of carbon dioxide will 4 moles of lithium hydroxide (lioh) absorb? (one mole of any gads occupies 22.4 l under certain conditions of temperature and pressure. assume those conditions for this equation.) 45l 6.0l 3.0l 34l
Answers: 1

Chemistry, 22.06.2019 14:00
How is the atomic number of a nucleus changed by alpha decay
Answers: 2

Chemistry, 22.06.2019 18:00
How does climate change cause the ocean's thermohaline current to slow down?
Answers: 3
You know the right answer?
Two moles of propene (c3h6) gas react with nine moles of oxygen (o2) gas to produce six moles of car...
Questions

English, 02.01.2020 18:31



Advanced Placement (AP), 02.01.2020 18:31



Engineering, 02.01.2020 18:31



Advanced Placement (AP), 02.01.2020 18:31



Mathematics, 02.01.2020 18:31

Computers and Technology, 02.01.2020 18:31

Computers and Technology, 02.01.2020 18:31


Computers and Technology, 02.01.2020 18:31



Computers and Technology, 02.01.2020 18:31

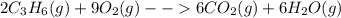
 to the gaseous state-of-matter at which the chemical reaction is carried out. Such coefficients are useful to respect the law of conservation of mass.
to the gaseous state-of-matter at which the chemical reaction is carried out. Such coefficients are useful to respect the law of conservation of mass.

