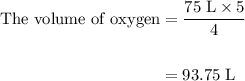Acetylene gas (c2h2) reacts with oxygen gas (o2) to produce carbon dioxide (co2) and water vapor (h2o). how many liters of c2h2 are required to produce 75.0 l of co2? l what volume of h2o is produced? l what volume of o2 is required? l when making the calculations, did you need to find the number of moles?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:00
Write a hypothesis that answers the lesson question, “while observing a chemical reaction, how can you tell which reactant is limiting? ” hypothesis: if a substance is the limiting reactant, then . . because . .
Answers: 1

Chemistry, 22.06.2019 09:00
Astudent is asked to identify and element that is pale yellow brittle solid and does not conduct electricity. at which location in this periodic table would the element most likely be found?
Answers: 2

Chemistry, 22.06.2019 14:00
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3

Chemistry, 22.06.2019 14:30
Which of the following represents the ester functional group? a. -coo- b. -cho c. -cooh d. c=o
Answers: 1
You know the right answer?
Acetylene gas (c2h2) reacts with oxygen gas (o2) to produce carbon dioxide (co2) and water vapor (h2...
Questions

Chemistry, 26.10.2019 10:43



History, 26.10.2019 10:43


History, 26.10.2019 10:43


Physics, 26.10.2019 10:43

Physics, 26.10.2019 10:43

Health, 26.10.2019 10:43

Mathematics, 26.10.2019 10:43

English, 26.10.2019 10:43


Geography, 26.10.2019 10:43



History, 26.10.2019 10:43


Mathematics, 26.10.2019 10:43

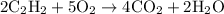
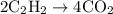
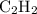 = 4 L of
= 4 L of 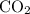
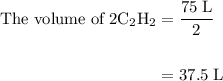
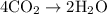
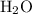
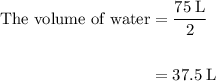
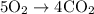
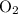 = 4 L of
= 4 L of