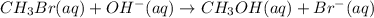Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 08:30
The characteristic of two different types of reactions are shown below. reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of and element. which statement is true about the atoms of the elements that participate in the two reactions? a: their identity changes in both reaction a and b. b: their identity changes in reaction a but not b. c: their identity changes in reaction b but not a. d: their identity remains the same.
Answers: 1

Chemistry, 22.06.2019 10:00
In a water molecule, hydrogen and oxygen are held together by a(an) bond. a) double covalent b) ionic c) nonpolar covalent d) hydrogen e) polar covalent
Answers: 1

Chemistry, 22.06.2019 12:30
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1

You know the right answer?
Which term describes this reaction? ch3br(aq) + oh-(aq) -> ch3oh(aq) + br-(aq)
a. additio...
a. additio...
Questions

Biology, 21.08.2019 07:30

Mathematics, 21.08.2019 07:30

Physics, 21.08.2019 07:30


Mathematics, 21.08.2019 07:30

History, 21.08.2019 07:30

Mathematics, 21.08.2019 07:30

Mathematics, 21.08.2019 07:30

Mathematics, 21.08.2019 07:30

Mathematics, 21.08.2019 07:30

Physics, 21.08.2019 07:30


Geography, 21.08.2019 07:30

Mathematics, 21.08.2019 07:30


Business, 21.08.2019 07:30

World Languages, 21.08.2019 07:30



Mathematics, 21.08.2019 07:30