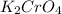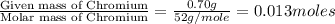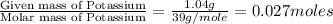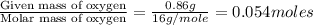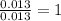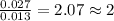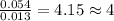Chemistry, 28.07.2019 00:30 lestessanders02
Analysis of a compound indicates that it contains 1.04 grams k 0.70 g cr and 0.86 g o. find its empirical formula

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:00
An excess of lithium oxide undergoes a synthesis reaction with water to produce lithium hydroxide li2o+h2o→2lioh if 1.05 g of water reacted, what is the theoretical yield of lithium hydroxide? a) 5.83 x 10–2 g lioh b) 1.17 x 10–1 g lioh c) 2.79 x 100 g lioh d) 1.40 x 100 g lioh
Answers: 1


Chemistry, 22.06.2019 18:00
Answer asap need to be answered by wednesday morning explain how a buffer works, using an ethanoic acid / sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 3

Chemistry, 23.06.2019 20:30
If 4.88 grams of zn react with 5.03 grams of s8 to produce 6.02 grams of zns, what are the theoretical yield and percent yield of this reaction? be sure to show the work that you did to solve this problem.unbalanced equation: zn + s8 yields zns
Answers: 3
You know the right answer?
Analysis of a compound indicates that it contains 1.04 grams k 0.70 g cr and 0.86 g o. find its empi...
Questions



Computers and Technology, 09.07.2020 02:01

Business, 09.07.2020 02:01








English, 09.07.2020 02:01



Mathematics, 09.07.2020 02:01

Physics, 09.07.2020 02:01


Mathematics, 09.07.2020 02:01


History, 09.07.2020 02:01