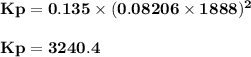Chemistry, 28.07.2019 07:00 ismailear18
For the reaction 2ch4(g)⇌c2h2(g)+3h2(g) k = 0.135 at 1615 ∘c . what is kp for the reaction at this temperature?

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 03:30
If a solution is considered basic, then a) the hydroxide ion and hydronium ion concentrations are equal. b) the hydroxide ion concentration is less than the hydronium ion concentration. c) the hydronium ion concentration is greater than the hydroxide ion concentration. d) the hydroxide ion concentration is greater than the hydronium ion concentration.
Answers: 1

Chemistry, 22.06.2019 06:00
One does not belong why? ice, gold ,wood ,diamond and table salt
Answers: 1

Chemistry, 22.06.2019 10:50
Someone offer some answers to this, i will give 98 coins and mark as brainliest! i will put the rest of the lab down in the comments,solutions pre-lab questions: in this lab, you will make fruit drinks with powdered drink mix. complete the pre-lab questions to get the values you need for your drink solutions. calculate the molar mass of powered fruit drink mix, made from sucrose (c12h22o11).using stoichiometry, determine the mass of powdered drink mix needed to make a 1.0 m solution of 100 ml. (hint: use molarity = to find the moles of drink mix, then convert moles to grams using a mole conversion.)what mass of powdered drink mix is needed to make a 0.5 m solution of 100 ml?
Answers: 1
You know the right answer?
For the reaction 2ch4(g)⇌c2h2(g)+3h2(g) k = 0.135 at 1615 ∘c . what is kp for the reaction at this t...
Questions

Mathematics, 31.10.2020 01:00

Mathematics, 31.10.2020 01:00


English, 31.10.2020 01:00

Mathematics, 31.10.2020 01:00


History, 31.10.2020 01:00


Social Studies, 31.10.2020 01:00

Biology, 31.10.2020 01:00

Biology, 31.10.2020 01:00

Mathematics, 31.10.2020 01:00


Mathematics, 31.10.2020 01:00

Physics, 31.10.2020 01:00



English, 31.10.2020 01:00

History, 31.10.2020 01:00

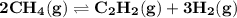
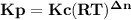
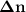 - moles of gaseous products = 1+3-2 = 2
- moles of gaseous products = 1+3-2 = 2