
Chemistry, 29.07.2019 16:30 angelinacortes11
Consider the unbalanced equation for the combustion of hexane: αc6h14(g)+βo2(g)→γco2(g)+δh2o(g) part a balance the equation. give your answer as an ordered set of numbers α, β, γ, use the least possible integers for the coefficients. α, β, γ, δ = request answer part b determine how many moles of o2 are required to react completely with 6.4 moles c6h14. express your answer using two significant figures. n = mol

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 21:30
Calculate the h3o+ concentration in a solution of acetic acid if the concentration of molecular acetic acid present at equilibrium is 9.97x10^-3 m and k for the dissociation is 1.86x10^-5. ch3cooh(aq)+h2o(> h3o^+(aq)+ch3coo^-(aq)
Answers: 2



Chemistry, 23.06.2019 10:00
Lord kelvin described the concept of absolute zero temperature and the laws relating to the change of thermal energy during chemical reactions what type of chemist would he be considered today
Answers: 1
You know the right answer?
Consider the unbalanced equation for the combustion of hexane: αc6h14(g)+βo2(g)→γco2(g)+δh2o(g) par...
Questions

History, 17.03.2021 23:50





History, 17.03.2021 23:50



Mathematics, 17.03.2021 23:50




Physics, 17.03.2021 23:50

Chemistry, 17.03.2021 23:50

Physics, 17.03.2021 23:50


Computers and Technology, 17.03.2021 23:50



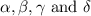 are 2, 19, 12 and 14 respectively.
are 2, 19, 12 and 14 respectively.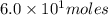 of
of 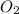 are required to react with 6.4 moles of
are required to react with 6.4 moles of 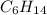 .
.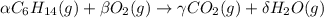
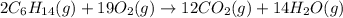
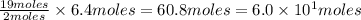 of
of 

