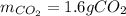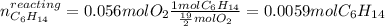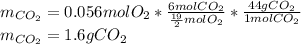Problem page liquid hexane ch3ch24ch3 will react with gaseous oxygen o2 to produce gaseous carbon dioxide co2 and gaseous water h2o . suppose 1.72 g of hexane is mixed with 1.8 g of oxygen. calculate the maximum mass of carbon dioxide that could be produced by the chemical reaction. be sure your answer has the correct number of significant digits.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 21:20
If a simple machine aduces the strength of a force, what must be increased? the speed of the input force the work the simple machine performs the size of the simple machine the distance over which the force is applied
Answers: 1

Chemistry, 22.06.2019 23:00
What is formed when amino acids form long chains or polymerize
Answers: 1

Chemistry, 23.06.2019 05:00
How is electrolysis most commonly used to produce an energy source? a - splitting water molecules produces oxygen, which organisms breathe to fuel their bodies. b - splitting water molecules produces hydrogen gas, which is used to power machines through hydrogen fuel cells. c - splitting carbon dioxide molecules produces coal, a form of carbon that can be burned to produce heat. d - splitting carbon dioxide molecules produces natural gas, which can be burned to generate electricity in power plants.
Answers: 1
You know the right answer?
Problem page liquid hexane ch3ch24ch3 will react with gaseous oxygen o2 to produce gaseous carbon di...
Questions

English, 22.10.2020 08:01





Mathematics, 22.10.2020 08:01


Physics, 22.10.2020 08:01


Biology, 22.10.2020 08:01


Health, 22.10.2020 08:01


Health, 22.10.2020 08:01



Mathematics, 22.10.2020 08:01

History, 22.10.2020 08:01

Geography, 22.10.2020 08:01

English, 22.10.2020 08:01