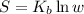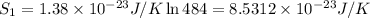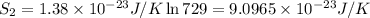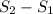Chemistry, 01.08.2019 09:00 22moneymorgan
An oxygen o2 molecule is adsorbed on a patch of surface (see sketch at right). this patch is known to contain 484 adsorption sites. the o2 molecule has enough energy to move from site to site, so it could be on any one of them. suppose additional surface becomes exposed, so that 729 adsorption sites are now available for the molecule. calculate the change in entropy. round your answer to 3 significant digits, and be sure it has the correct unit symbol.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:40
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3



Chemistry, 22.06.2019 13:30
Which statements are true concerning mineral formation? check all that apply. the slower the cooling, the larger the crystals. the faster the cooling, the smaller the crystals. crystals formed from magma are smaller than crystals formed from lava. minerals can only form in solutions when the solution is heated deep underground. when a solution cools, elements and compounds leave the solution and crystallize as minerals. minerals formed from hot water solutions can form narrow channels in the surrounding rock.
Answers: 1
You know the right answer?
An oxygen o2 molecule is adsorbed on a patch of surface (see sketch at right). this patch is known t...
Questions




English, 28.02.2021 14:00


Geography, 28.02.2021 14:00

Mathematics, 28.02.2021 14:00




History, 28.02.2021 14:00


French, 28.02.2021 14:00




Mathematics, 28.02.2021 14:00

History, 28.02.2021 14:00

Mathematics, 28.02.2021 14:00

Physics, 28.02.2021 14:00

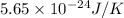 .
.