
Chemistry, 01.08.2019 10:00 journey888
Asubstance is known to contain 2.00 g of carbon for every 5.33 g of oxygen. how many grams of carbon will be present in 55.0 g of this substance?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 11:00
As air becomes more dense, (select all that apply) o. air weighs less o. gas molecules are closer together o. air is colder o. air weighs more o. gas molecules are further apart o. air is hotter
Answers: 3

Chemistry, 23.06.2019 00:20
Steam reforming of methane ( ch4) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. an industrial chemist studying this reaction fills a 1.5 l flask with 3.5 atm of methane gas and 1.3 atm of water vapor at 43.0°c. he then raises the temperature, and when the mixture has come to equilibrium measures the partial pressure of carbon monoxide gas to be 1 .0 atm. calculate the pressure equilibrium constant for the steam reforming of methane at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 1

Chemistry, 23.06.2019 00:30
Gasoline has a density of 0.740 g/ml. if you have 328 grams of gasoline, what is the volume in milliliters?
Answers: 1

Chemistry, 23.06.2019 01:00
Na chemical reaction, activation energy increases the of the reactants. this outcome causes the particles to collide, which results in the of new products.
Answers: 2
You know the right answer?
Asubstance is known to contain 2.00 g of carbon for every 5.33 g of oxygen. how many grams of carbon...
Questions



Social Studies, 15.12.2019 12:31


Social Studies, 15.12.2019 12:31


English, 15.12.2019 12:31


Biology, 15.12.2019 12:31

Mathematics, 15.12.2019 12:31


Mathematics, 15.12.2019 12:31



History, 15.12.2019 12:31




Mathematics, 15.12.2019 12:31

Mathematics, 15.12.2019 12:31

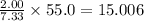 grams of carbon.
grams of carbon.

