

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:10
Explain the relationship between forward and backward reactions in equilibrium, and predict how changing the amount of a reactant (creating a tension) will affect that relationship.
Answers: 1

Chemistry, 22.06.2019 07:00
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

Chemistry, 22.06.2019 22:00
Does the number of ions in solution increase, decrease, or remain constant? it continuously decreases. it continuously increases. it decreases at first, then increases. it increases at first, then decreases.
Answers: 3

Chemistry, 23.06.2019 00:00
#7 how does the structure of amino acids allow them to form a polypeptide? each amino acid has an amino group and a carboxyl group. each amino acid has a hydrogen atom and a carboxyl group. each amino acid has a carboxyl group and an r group. each amino acid has an r group and a hydrogen atom.
Answers: 1
You know the right answer?
If a sample of pure silver (ag) contains 3.35 x 1022 atoms of silver, how many moles of silver are i...
Questions

Mathematics, 07.04.2021 22:10

Mathematics, 07.04.2021 22:10

Health, 07.04.2021 22:10

Mathematics, 07.04.2021 22:10


Chemistry, 07.04.2021 22:10




English, 07.04.2021 22:10

Mathematics, 07.04.2021 22:10

Mathematics, 07.04.2021 22:10






Mathematics, 07.04.2021 22:10


Mathematics, 07.04.2021 22:10

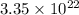
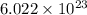 number of atoms.
number of atoms. number of atoms occupies 1 mole of a element.
number of atoms occupies 1 mole of a element.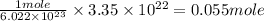 of silver element.
of silver element.

