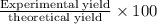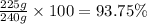In the manufacturing process of sulfuric acid, sulfur dioxide is reacted with oxygen to produce sulfur trioxide. using the equation, 2so2 (g) + o2 imported asset 2so3 (g), if 192 g of sulfur dioxide is given the opportunity to react with an excess of oxygen to produce 225 g of sulfur trioxide, what is the percent yield of this reaction?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:00
Which best describes how johannes kepler developed his laws of planetary motion
Answers: 3

Chemistry, 23.06.2019 05:10
Will mark as brainliest ! how many grams of iron metal do you expect to be produced when 245 grams of an 80.5 percent by mass iron (ii) nitrate solution react with excess aluminum metal? show all work needed to solve this problem!
Answers: 1

Chemistry, 23.06.2019 08:00
Which of the following notations would be the appropriate and final way to display the formula for magnesium chloride a. mgcl2 b. mg+2cl–1 c. mgcl2 d. mgcl
Answers: 2

You know the right answer?
In the manufacturing process of sulfuric acid, sulfur dioxide is reacted with oxygen to produce sulf...
Questions







Biology, 20.07.2019 12:00

Social Studies, 20.07.2019 12:00

Biology, 20.07.2019 12:00








Chemistry, 20.07.2019 12:00



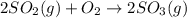
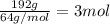
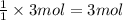 of sulfur trioxide
of sulfur trioxide