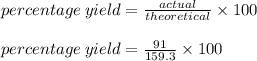Chemistry, 02.08.2019 16:00 slhfbfjcys
What is the percent yield of a reaction in which 200. g of phosphorus trichloride reacts with excess water to form 91.0 g of hcl and aqueous phosphorous acid (h3po3)?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.036m naoh best answer will be brainliest
Answers: 3

Chemistry, 22.06.2019 17:00
What is the approximate vapor pressure when the gas condenses at 70 degrees celsius
Answers: 2

Chemistry, 22.06.2019 21:30
While in europe, if you drive 125 km per day, how much money would you spend on gas in one week if gas costs 1.10 euros per liter and your car’s gas mileage is 32.0 mi/gal? assume that 1 euro=1.26 dollars
Answers: 2

Chemistry, 23.06.2019 00:30
On the periodic table, elements are arranged by which of the following. a. mass numbers. b. increasing atomic number. c. alphabetical order. or d. density
Answers: 1
You know the right answer?
What is the percent yield of a reaction in which 200. g of phosphorus trichloride reacts with excess...
Questions

Mathematics, 26.01.2021 08:30





Mathematics, 26.01.2021 08:30



Mathematics, 26.01.2021 08:30

Mathematics, 26.01.2021 08:30




SAT, 26.01.2021 08:30

Business, 26.01.2021 08:30

Mathematics, 26.01.2021 08:30




Chemistry, 26.01.2021 08:30