Chemistry, 03.08.2019 05:00 ahmedeldyame
Which of the following will favor a spontaneous change? 1.+δh. 2.−δh. 3.+δs. 4.−δs.

Answers: 1


Another question on Chemistry



Chemistry, 22.06.2019 18:10
Consider the following reaction at equilibrium: c(s)+h2o(g)⇌co(g)+h2(g) predict whether the reaction will shift left, shift right, or remain unchanged upon each of the following disturbances. a) c is added to the reaction mixture. b) h2ois condensed and removed from the reaction mixture c) co is added to the reaction mixture d) h2 is removed from the reaction mixture.
Answers: 3

Chemistry, 22.06.2019 19:20
Consider hydrogen in an excited state n = 5n=5 that emits photons to reach the ground state. there are various possible transitions other than straight to the ground state that can occur; for example, it can drop to the n = 3n=3 state followed by the n = 3n=3 to the ground state transition. which of the possible transitions will result in the emission of a photon in the visible region?
Answers: 3
You know the right answer?
Which of the following will favor a spontaneous change? 1.+δh. 2.−δh. 3.+δs. 4.−δs....
Questions






English, 01.05.2021 05:10




Mathematics, 01.05.2021 05:10



Physics, 01.05.2021 05:10

Mathematics, 01.05.2021 05:10


Geography, 01.05.2021 05:10

Chemistry, 01.05.2021 05:10


Chemistry, 01.05.2021 05:10

Mathematics, 01.05.2021 05:10

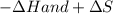 will favor a spontaneous change
will favor a spontaneous change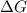 (Change in gibbs free enegy) should be negative.
(Change in gibbs free enegy) should be negative. 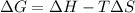
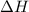 is change in enthalpy for reaction, T is temperature in kelvin scale and
is change in enthalpy for reaction, T is temperature in kelvin scale and 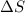 is change in entropy for reaction.
is change in entropy for reaction.