
Chemistry, 03.08.2019 07:30 2021CanadyRaniya
The combustion of 14 grams of co, according to the reaction co(g) + ½o2(g) → co2(g) + 67.6 kcal, gives off how much heat?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:00
For ai it's atomic number is 13 and it's mass number is 27 how many neutrons does it have
Answers: 1

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 06:00
Which of the following did jj thompson discover about atoms? a)an atom has an internal structure. b) atoms are tiny indivisible particles. c)electrons orbit the nucleus of an atom. d) the nucleus of an atom contains protons and neutrons.
Answers: 2

Chemistry, 22.06.2019 06:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 3
You know the right answer?
The combustion of 14 grams of co, according to the reaction co(g) + ½o2(g) → co2(g) + 67.6 kcal, giv...
Questions

Mathematics, 26.01.2022 18:00


Mathematics, 26.01.2022 18:00





Mathematics, 26.01.2022 18:00






Computers and Technology, 26.01.2022 18:10


Business, 26.01.2022 18:10


History, 26.01.2022 18:10


Social Studies, 26.01.2022 18:10

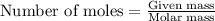
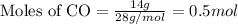
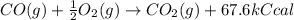
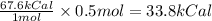 of heat.
of heat.

