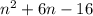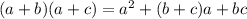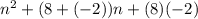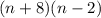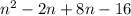Chemistry, 28.11.2019 11:31 dinadenoirefan
Find the product.
(n + 8)(n - 2)
n2- 6n - 16
n2+ 6n - 16
n2- 6n + 16

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:30
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 22.06.2019 09:00
What is the percentage composition of carbon in the compound ch4
Answers: 1

Chemistry, 22.06.2019 16:30
At 20°c, a sample of h2o liquid and a sample of co2 gas each have the same average kinetic energy. why is one a liquid and the other a gas at this temperature?
Answers: 1

Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1
You know the right answer?
Find the product.
(n + 8)(n - 2)
n2- 6n - 16
n2+ 6n - 16
n2- 6n + 16...
(n + 8)(n - 2)
n2- 6n - 16
n2+ 6n - 16
n2- 6n + 16...
Questions

Mathematics, 25.06.2019 22:10






History, 25.06.2019 22:20





Social Studies, 25.06.2019 22:20

English, 25.06.2019 22:20