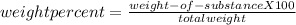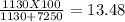Making homemade ice cream is one of life's great pleasures. fresh milk and cream, sugar, and flavorings are churned in a bucket suspended in an ice–water mixture, the freezing point of which has been lowered by adding rock salt. one manufacturer of home ice cream freezers recommends adding 2.50 lb (1130 g) of rock salt (nacl) to 16.0 lb of ice (7250 g) in a 4-qt freezer. calculate the weight percent of nacl of the solution that will result when this mixture melts.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 18:00
During which movies do spring tides new moon first quarter waxing gibbous waxing
Answers: 1


Chemistry, 22.06.2019 10:00
Americium-241 undergoes fission to produce three neutrons per fission event. if a neutron-absorbing material is mixed in with this sample so that the rate of neutron production drops down to 1.8 neutrons per fission event, which will be effective at achieving a critical mass? check all that apply. remove a deflective shield surrounding the sample. remove absorbent material mixed in with the sample. compress the sample of americium-241.
Answers: 1

Chemistry, 22.06.2019 10:40
Which buffer would be better able to hold a steady ph on the addition of strong acid, buffer 1 or buffer 2? explain. buffer 1: a solution containing 0.10 m nh4cl and 1 m nh3. buffer 2: a solution containing 1 m nh4cl and 0.10 m nh3
Answers: 1
You know the right answer?
Making homemade ice cream is one of life's great pleasures. fresh milk and cream, sugar, and flavori...
Questions


Biology, 24.05.2021 18:10

Mathematics, 24.05.2021 18:10

English, 24.05.2021 18:10


English, 24.05.2021 18:10


Mathematics, 24.05.2021 18:10


Chemistry, 24.05.2021 18:10

Chemistry, 24.05.2021 18:10

Computers and Technology, 24.05.2021 18:10

Chemistry, 24.05.2021 18:10

Mathematics, 24.05.2021 18:10

English, 24.05.2021 18:10

Mathematics, 24.05.2021 18:10


Mathematics, 24.05.2021 18:10

Mathematics, 24.05.2021 18:10