
Chemistry, 27.07.2019 16:50 fernandezvela27
When converting a number from scientific notation to standard notation if the power of 10 is __ move the decimal place to the left

Answers: 1


Another question on Chemistry

Chemistry, 20.06.2019 18:02
This is the star at the center of the solar system around which the earth and other matter orbit
Answers: 1

Chemistry, 21.06.2019 17:10
For which one of the following reactions is the value of δh° rxn equal to δh° f for the product? a. 2 h2 (g) + o2 (g) → 2 h2o (l) b. n2 (g) + o2 (g) → 2 no (g) c. 2 h2 (g) + o2 (g) → 2 h2o (g) d. h2o (l) + 1/2 o2 (g) → h2o2 (l) e. none of the above
Answers: 1

Chemistry, 22.06.2019 05:50
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2
You know the right answer?
When converting a number from scientific notation to standard notation if the power of 10 is __ move...
Questions



Computers and Technology, 21.01.2021 04:30

Mathematics, 21.01.2021 04:30

Mathematics, 21.01.2021 04:30

Mathematics, 21.01.2021 04:30

Mathematics, 21.01.2021 04:30

Mathematics, 21.01.2021 04:30



Mathematics, 21.01.2021 04:30

Mathematics, 21.01.2021 04:30



Mathematics, 21.01.2021 04:30

Mathematics, 21.01.2021 04:30

Mathematics, 21.01.2021 04:30

Mathematics, 21.01.2021 04:30

History, 21.01.2021 04:30

History, 21.01.2021 04:30


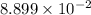
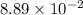 this is written in the scientific notation and the standard notation of this number will be, 0.00889. That means, when the power of 10 is negative then the decimal moves to the left side.
this is written in the scientific notation and the standard notation of this number will be, 0.00889. That means, when the power of 10 is negative then the decimal moves to the left side.

