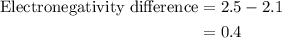Chemistry, 20.07.2019 19:00 jadajohnson14
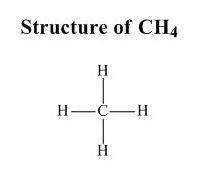
Which statement explains why a molecule of ch4 is nonpolar? (1) the bonds between the atoms in a ch4 molecule are polar. (2) the bonds between the atoms in a ch4 molecule are ionic. (3) the geometric shape of a ch4 molecule distributes the charges symmetrically. (4) the geometric shape of a ch4 molecule distributes the charges asymmetrically.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:00
2h2s + 3o2 2so2 + 2h2o which option gives the correct mole ratios? h2s: so2 = 2: 2 and o2: h2o = 3: 2 h2s: so2 = 2: 3 and o2: h2o = 3: 2 h2s: so2 = 4: 4 and o2: h2o = 5: 4 h2s: so2 = 4: 6 and o2: h2o = 4: 4
Answers: 1

Chemistry, 22.06.2019 00:40
1) in saturated limewater, [h+ ]=3.98x10-13 m. a) find [oh]-/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 2) in butter, [h+ ]=6.0x10-7 m. a) find [oh]-/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 3) in peaches, [oh]=3.16x10-11 m a) find [h+ ]/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 4) during the course of the day, human saliva varies between being acidic and basic. if [oh]=3.16x10-8 m, a) find [h+ ]/ b) what is the ph? / c) is the solution acidic, basic, or neutral? /
Answers: 3

Chemistry, 22.06.2019 10:50
Someone offer some answers to this, i will give 98 coins and mark as brainliest! i will put the rest of the lab down in the comments,solutions pre-lab questions: in this lab, you will make fruit drinks with powdered drink mix. complete the pre-lab questions to get the values you need for your drink solutions. calculate the molar mass of powered fruit drink mix, made from sucrose (c12h22o11).using stoichiometry, determine the mass of powdered drink mix needed to make a 1.0 m solution of 100 ml. (hint: use molarity = to find the moles of drink mix, then convert moles to grams using a mole conversion.)what mass of powdered drink mix is needed to make a 0.5 m solution of 100 ml?
Answers: 1

Chemistry, 22.06.2019 13:50
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3
You know the right answer?
Which statement explains why a molecule of ch4 is nonpolar? (1) the bonds between the atoms in a ch...
Questions




Mathematics, 06.05.2020 03:20

History, 06.05.2020 03:20


Mathematics, 06.05.2020 03:20

Mathematics, 06.05.2020 03:20


Mathematics, 06.05.2020 03:20

Mathematics, 06.05.2020 03:20

Mathematics, 06.05.2020 03:20


Mathematics, 06.05.2020 03:20




Mathematics, 06.05.2020 03:20

Physics, 06.05.2020 03:20

Mathematics, 06.05.2020 03:20

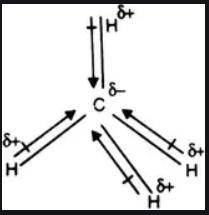
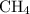 is nonpolar is as follows:
is nonpolar is as follows:
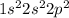 . It requires four electrons to complete its octet. The atomic number of hydrogen is 1 and its electronic configuration is
. It requires four electrons to complete its octet. The atomic number of hydrogen is 1 and its electronic configuration is 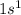 . Each carbon requires four electrons to complete its octet so it forms four covalent bonds with four hydrogen atoms and one molecule of
. Each carbon requires four electrons to complete its octet so it forms four covalent bonds with four hydrogen atoms and one molecule of 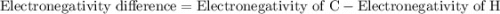 …… (1)
…… (1)