Chemistry, 19.07.2019 17:20 Raekwon3232
Determine the limiting reactant (lr) and the mass (in g) of nitrogen that can be formed from 50.0 g n2o4 and 45.0 g n2h4. some possibly useful molar masses are as follows: n2o4 = 92.02 g/mol, n2h4 = 32.05 g/mol. n2o4(l) + 2 n2h4(l) → 3 n2(g) + 4 h2o(g) determine the limiting reactant (lr) and the mass (in g) of nitrogen that can be formed from 50.0 g n2o4 and 45.0 g n2h4. some possibly useful molar masses are as follows: n2o4 = 92.02 g/mol, n2h4 = 32.05 g/mol. n2o4(l) + 2 n2h4(l) → 3 n2(g) + 4 h2o(g) lr = n2o4, 105 g n2 formed lr = n2h4, 59.0 g n2 formed lr = n2o4, 45.7 g n2 formed no lr, 45.0 g n2 formed lr = n2h4, 13.3 g n2 formed

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 19:30
The crust of earth may a- continets and ocean floors. b-continents only. c-layers of sedimentary rocks and continents. d-all of the above
Answers: 2

Chemistry, 22.06.2019 02:00
Which of the following is not a good technique for managing used oil? a) have specific, labeled catch pans available for technicians who are collecting oil b) spills in your shop and any releases on pavement or outside should be poured down a drain c) do not use oil containers for antifreeze or other non-similar fluids d) be prepared for oil spills with the proper absorbents
Answers: 1


You know the right answer?
Determine the limiting reactant (lr) and the mass (in g) of nitrogen that can be formed from 50.0 g...
Questions





Mathematics, 28.11.2019 08:31


History, 28.11.2019 08:31



English, 28.11.2019 08:31

Mathematics, 28.11.2019 08:31

Mathematics, 28.11.2019 08:31



Mathematics, 28.11.2019 08:31



World Languages, 28.11.2019 08:31

Advanced Placement (AP), 28.11.2019 08:31

Mathematics, 28.11.2019 08:31

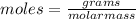 (molar mass of N2= 28)
(molar mass of N2= 28)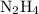 . The moles of nitrogen formed has been 2.1 mol of nitrogen.
. The moles of nitrogen formed has been 2.1 mol of nitrogen. and
and