

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 08:30
7. using your knowledge of colligative properties explain whether sodium chloride or calcium chloride would be a more effective substance to melt the ice on a slick sidewalk. use 3 – 4 sentences in your explanation. 8. when a 2.5 mol of sugar (c12h22o11) are added to a certain amount of water the boiling point is raised by 1 celsius degree. if 2.5 mol of aluminum nitrate is added to the same amount of water, by how much will the boiling point be changed? show all calculations leading to your answer or use 3 – 4 sentences to explain your answer. 9. if 5.40 kcal of heat is added to 1.00 kg of water at 100⁰c, how much steam at 100⁰c is produced? show all calculations leading to an answer. 10. the freezing of water at 0⁰c can be represented as follows: h2o (l) ↔ h2o(s) the density of liquid water is 1.00 g/cm3. the density of ice is 0.92 g/cm3. in 3 – 4 sentences explain why applying pressure causes ice to melt.
Answers: 1

Chemistry, 22.06.2019 09:00
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1


Chemistry, 22.06.2019 22:30
Which of the following is true about the speed of light? it depends on the wavelength.
Answers: 3
You know the right answer?
What are the relative numbers of h3o+ and oh- ions in an acidic and alkaline and a neutral solution?...
Questions


History, 07.07.2019 15:00

Mathematics, 07.07.2019 15:00

Spanish, 07.07.2019 15:00

Chemistry, 07.07.2019 15:00

Spanish, 07.07.2019 15:00

History, 07.07.2019 15:00

Spanish, 07.07.2019 15:00


Spanish, 07.07.2019 15:00

History, 07.07.2019 15:00


English, 07.07.2019 15:00


Spanish, 07.07.2019 15:00

Computers and Technology, 07.07.2019 15:00


French, 07.07.2019 15:00


Business, 07.07.2019 15:00

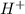 ) or hydronium (
) or hydronium (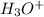 ) ions as compared to hydroxide ions (
) ions as compared to hydroxide ions (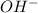 ).
). ![[H^{+}] or [H_{3}O^{+}]](/tpl/images/0100/3599/e5aac.png) >
> ![[OH^{-}]](/tpl/images/0100/3599/e46dd.png)


