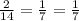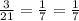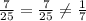Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:00
Cucl2 + 2nano3 cu(no3)2 + 2nacl what is the percent yield of nacl if 31.0 g of cucl2 reacts with excess nano3 to produce 21.2 g of nacl? 49.7% 58.4% 63.6% 78.7%
Answers: 1

Chemistry, 23.06.2019 03:00
Is it safe to take 450mg of diphenhydramine hydrochloride?
Answers: 1

Chemistry, 23.06.2019 23:00
2.50 mol nocl was placed in a 2.50 l reaction vessel at 400ºc. after equilibrium was established, it was found that 28% of the nocl had dissociated according to the equation 2nocl(g) picture 2no(g) + cl2(g). calculate the equilibrium constant, kc, for the reaction.
Answers: 2

Chemistry, 23.06.2019 23:10
Exactly 1.00 ml of an aqueous solution of hno3 is diluted to 100.0 ml. it takes 29.78 ml of 0.0142 m ca(oh)2 to convert all of the hno3 to ca(no3)2. the other product of the reaction is water. calculate the molarity of the undiluted hno3 solution. petrucci, ralph h.. general chemistry (p. 144). pearson education. kindle edition.
Answers: 2
You know the right answer?
Use common factors to write 2 fractions equivalent to 6/42. a) 4/16,2/21 b) 2/14,3/21 c) 2/14,4/16 d...
Questions

Physics, 21.10.2021 16:30



Chemistry, 21.10.2021 16:40


SAT, 21.10.2021 16:40



Mathematics, 21.10.2021 16:40

SAT, 21.10.2021 16:40


Mathematics, 21.10.2021 16:40

Business, 21.10.2021 16:40



Mathematics, 21.10.2021 16:40


History, 21.10.2021 16:40


Mathematics, 21.10.2021 16:40

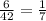
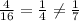
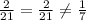
 .
.