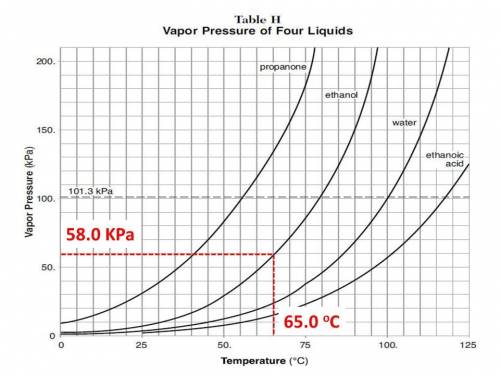
Chemistry, 13.07.2019 05:50 penaabel1492
At 65 degrees celsius, which compound has a vapor pressure of 58 kilopascals? 1. ethanoic acid2. ethanol 3. propanone 4. water

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 15:50
In a popular classroom demonstration, solid sodium is added to liquid water and reacts to produce hydrogen gas and aqueous sodium hydroxide. part a write a balanced chemical equation for this reaction. express your answer as a chemical equation. identify all of the phases in your answer.
Answers: 3

Chemistry, 22.06.2019 09:00
Look at the spectrums of a star moving towards earth and a motionless star. which of these is a correct inference that can be draw from the observation of the two spectrums? (2 points) the spectrum of a motionless star is difficult to be viewed separately using oridinary telescopes. the spectrum of a motionless star is identical to the spectrum of a star which moves towards earth. the spectrum of a star shifts towards the red region when the star moves towards earth. the spectrum of a star shifts towards the blue region when the star moves towards earth.
Answers: 2

Chemistry, 22.06.2019 14:00
Calculate the energy required to ionize a hydrogen atom to an excited state where the electron is initially in the n = 5 energy level. report your answer in kilojoules
Answers: 1

Chemistry, 23.06.2019 15:30
Acontainer holds 6.4 moles of gas. hydrogen gas makes up 25% of the total moles in the container. if the total pressure is 1.24atm. what is the partial pressure of hydrogen
Answers: 3
You know the right answer?
At 65 degrees celsius, which compound has a vapor pressure of 58 kilopascals? 1. ethanoic acid2. et...
Questions


Mathematics, 22.01.2021 20:00

Mathematics, 22.01.2021 20:00

Health, 22.01.2021 20:00

Mathematics, 22.01.2021 20:00

Mathematics, 22.01.2021 20:00


Biology, 22.01.2021 20:00

Mathematics, 22.01.2021 20:00

Mathematics, 22.01.2021 20:00




Mathematics, 22.01.2021 20:00



Computers and Technology, 22.01.2021 20:00

English, 22.01.2021 20:00


Computers and Technology, 22.01.2021 20:00