Chemistry, 10.07.2019 02:50 lightskinbaby2
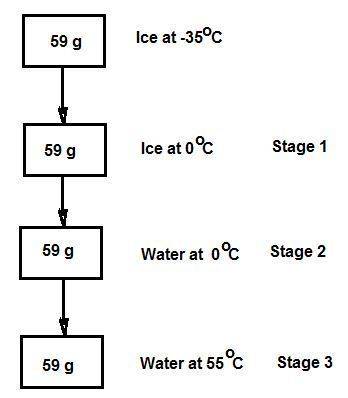
Consider the specific heats h2o(s) = 2.09 j/g · ◦c, h2o (ℓ) = 4.18 j/g · ◦c, and h2o(g) = 2.03 j/g · ◦c. the heat of fusion for water is 334 j/g and its heat of vaporization is 2260 j/g. calculate the amount of heat required to convert 59 g of ice at −35◦c completely to liquid water at 55◦c. answer in units of kj.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:30
If i make a solution by adding 83grams of sodium hydroxide to 750ml i’d water what is the molarity of sodium hydroxide
Answers: 1


Chemistry, 22.06.2019 06:30
Identify the missing numbers below to show the result of multiplying the numbers (1.6 × 10-19)(5.0 × 106) = c × 10d
Answers: 1

Chemistry, 23.06.2019 07:00
Which of the following statements is true? an atom consists of protons, electrons, and neutrons.an atom consists of protons and neutrons.an atom consists of electrons bonded to one another.an atom consists of protons bonded to one another.
Answers: 1
You know the right answer?
Consider the specific heats h2o(s) = 2.09 j/g · ◦c, h2o (ℓ) = 4.18 j/g · ◦c, and h2o(g) = 2.03 j/g ·...
Questions


Computers and Technology, 21.10.2020 08:01





Mathematics, 21.10.2020 08:01


Mathematics, 21.10.2020 08:01

English, 21.10.2020 08:01

Mathematics, 21.10.2020 08:01


Computers and Technology, 21.10.2020 08:01

Mathematics, 21.10.2020 08:01

Mathematics, 21.10.2020 08:01


Computers and Technology, 21.10.2020 08:01

English, 21.10.2020 08:01

Mathematics, 21.10.2020 08:01

Mathematics, 21.10.2020 08:01