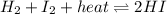Chemistry, 09.07.2019 19:00 tddreviews
Consider the chemical reaction in equilibrium. h2 + i2 + heat ⬄ 2hi what will happen to the chemical equilibrium if the temperature of the system is increased? a)the direction of the chemical equilibrium will shift to the right, favoring the forward reaction. b)the chemical equilibrium will not be affected by an increase in temperature. c)the direction of the chemical equilibrium will shift to the left, favoring the reverse reaction. d) the chemical equilibrium will be lost permanently with a change of temperature.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 12:00
Give the set of reactants (including an alkyl halide and a nucleophile) that could be used to synthesize the following ether: draw the molecules on the canvas by choosing buttons from the tools (for bonds and charges), atoms, and templates toolbars, including charges where needed. ch3ch2och2ch2chch3 | ch3
Answers: 1

Chemistry, 22.06.2019 16:40
The diagram below shows the movement of particles. what does this piece of evidence best support? the collision theory the maxwell-boltzmann distribution the effect of pressure on reaction rates the effect of temperature on reaction rates
Answers: 3

Chemistry, 22.06.2019 23:30
The ammonia molecule in the diagram has the observed bond orientation because
Answers: 1

You know the right answer?
Consider the chemical reaction in equilibrium. h2 + i2 + heat ⬄ 2hi what will happen to the chemic...
Questions


English, 05.02.2021 09:20

Mathematics, 05.02.2021 09:20



English, 05.02.2021 09:20

Biology, 05.02.2021 09:20




Mathematics, 05.02.2021 09:20



World Languages, 05.02.2021 09:20


Social Studies, 05.02.2021 09:20


Mathematics, 05.02.2021 09:20