
Chemistry, 20.08.2019 00:30 manman1773
Question 1 if the enthalpy of a system increases, what value of δh describes the process? δh is positive. δh is negative. δh is zero δh increases.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 12:30
According to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? view available hint(s) according to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? trigonal bipyramidal tetrahedral square planar determination of electron geometry requires information on whether the electron groups are lone pairs or bonding groups.
Answers: 2

Chemistry, 22.06.2019 22:30
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3

Chemistry, 23.06.2019 11:20
Match each state of matter with the statement that best describes it.
Answers: 1

Chemistry, 23.06.2019 12:20
Amatch has about 21 milligrams of red phosphorus coating the tip. how many atoms of phosphorus is this?
Answers: 1
You know the right answer?
Question 1 if the enthalpy of a system increases, what value of δh describes the process? δh is pos...
Questions




Computers and Technology, 13.03.2020 17:59



Chemistry, 13.03.2020 17:59

Mathematics, 13.03.2020 17:59

World Languages, 13.03.2020 17:59





Mathematics, 13.03.2020 18:00

Mathematics, 13.03.2020 18:00




Arts, 13.03.2020 18:00

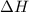 is positive.
is positive.![\Delta H_{rxn}=\sum [n\times \Delta H_{(product)}]-\sum [n\times \Delta H_{(reactant)}]](/tpl/images/0180/2055/42c4e.png)


