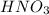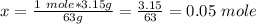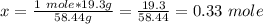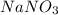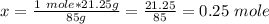Chemistry, 18.08.2019 19:30 tinyiaihfurlow
Calculate the number of moles 3.15 g of hno3 19.3 g of nacl 21.25 g of nano3

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Agood hypothesis includes which of the following? a: prediction b: data c: uncertainty d: conclusion
Answers: 1

Chemistry, 22.06.2019 04:40
Silver tarnishes as silver metal reacts with hydrogen sulfide, h2s, in the air. in this reaction, dark silver sulfide, au2s, covers the surface of silver. when silver is polished, this coating of silver sulfide can be removed from the surface. this makes the silver shiny again. enter the coefficients that balance the tarnishing reaction equation. (type 1 for no coefficient.)
Answers: 2

Chemistry, 22.06.2019 10:30
What woukd most likely be the transmittance at a 0.70 m solution of solute a? a) 7.6%b) 1.1%c)4.0%d)4.6%
Answers: 1

Chemistry, 22.06.2019 20:30
Consider the following unbalanced equation for the combustion of hexane: αc6h14(g)+βo2(g)→γco2(g)+δh2o(g) part a balance the equation. give your answer as an ordered set of numbers α, β, γ, use the least possible integers for the coefficients. α α , β, γ, δ = nothing request answer part b determine how many moles of o2 are required to react completely with 5.6 moles c6h14. express your answer using two significant figures. n n = nothing mol request answer provide feedback
Answers: 2
You know the right answer?
Calculate the number of moles 3.15 g of hno3 19.3 g of nacl 21.25 g of nano3...
Questions


Mathematics, 02.07.2019 15:00

Mathematics, 02.07.2019 15:00


Mathematics, 02.07.2019 15:00

Mathematics, 02.07.2019 15:00


History, 02.07.2019 15:00

English, 02.07.2019 15:00


Mathematics, 02.07.2019 15:00

Mathematics, 02.07.2019 15:00



English, 02.07.2019 15:00

Social Studies, 02.07.2019 15:00

English, 02.07.2019 15:00



Chemistry, 02.07.2019 15:00