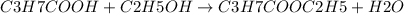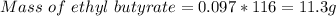Chemistry, 03.07.2019 17:00 maddyleighanne
Given 8.50 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100% yield?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:30
Choose all the answers that apply. as ocean depth increases, temperature decreases temperature increases pressure increases pressure decreases salinity increases density increases
Answers: 2

Chemistry, 23.06.2019 07:00
The image compares the arrangement of electrons in two different neutral atoms. a figure labeled atom q has a shaded sphere at the center of three concentric circles. the innermost circle has two black spheres. the middle circle has six black spheres. to the left of this figure is another figure labeled atom p. atom p has a shaded sphere at the center of three concentric circles. the innermost circle has two black spheres. the middle circle has seven black spheres. which of the following best explains the position of the two atoms in the periodic table?
Answers: 2

Chemistry, 23.06.2019 10:00
You dissolve 8.65 grams of lead(l) nitrate in water and then you add 2 50 grams of aluminum. this reaction occurs 2ai(s)+ 3pb(no3)2(aq) -3pb(s)+ 2aino3la(aq) the theoretical yield of solid lead?
Answers: 1

Chemistry, 23.06.2019 11:50
What is the oxidation half-reaction for this unbalanced redox equation? cr2o72– + fe2+ → cr3+ + fe3+ cr3+ → cr2o72– cr2o72– → cr3+ fe3+ → fe2+ fe2+ → fe3+?
Answers: 2
You know the right answer?
Given 8.50 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesi...
Questions




Mathematics, 22.11.2019 03:31

Mathematics, 22.11.2019 03:31


History, 22.11.2019 03:31



Mathematics, 22.11.2019 03:31


Mathematics, 22.11.2019 03:31

Mathematics, 22.11.2019 03:31


History, 22.11.2019 03:31

Mathematics, 22.11.2019 03:31


English, 22.11.2019 03:31

Mathematics, 22.11.2019 03:31