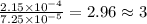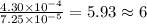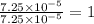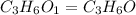Chemistry, 03.07.2019 11:10 AgentPangolin
(a) the characteristic odor of pineapple is due to ethyl butyrate, a compound containing carbon, hydrogen, and oxygen. combustion of 4.17 mg of ethyl butyrate produces 9.48 mg of co2 and 3.87 mg of h2o. what is the empirical formula of the compound?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:40
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2


Chemistry, 22.06.2019 18:30
Which rate indicates the number of children that would be born per woman if she were to live to the end of her child bearing years
Answers: 2

Chemistry, 23.06.2019 05:00
If 15 drops of ethanol from a medicine dropper weigh 0.60g, how many drops does it takes from a dropper to dispense 1.0ml of ethanol? density of ethanol is ethanol is 0.80g/ml.
Answers: 2
You know the right answer?
(a) the characteristic odor of pineapple is due to ethyl butyrate, a compound containing carbon, hyd...
Questions

Mathematics, 18.03.2021 02:50



Mathematics, 18.03.2021 02:50








Mathematics, 18.03.2021 02:50

English, 18.03.2021 02:50

Mathematics, 18.03.2021 02:50

Mathematics, 18.03.2021 02:50


Mathematics, 18.03.2021 02:50



Mathematics, 18.03.2021 02:50

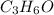
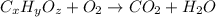
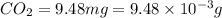
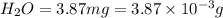
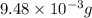 of carbon dioxide,
of carbon dioxide, 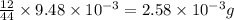 of carbon will be contained.
of carbon will be contained.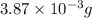 of water,
of water, 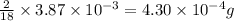 of hydrogen will be contained.
of hydrogen will be contained.![(4.17\times 10^{-3})-[(2.58\times 10^{-3})+(4.30\times 10^{-4})]=1.16\times 10^{-3}g](/tpl/images/0046/1661/df7a2.png)
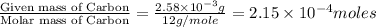
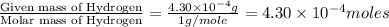
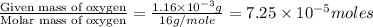
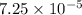 moles.
moles.