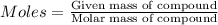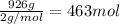Chemistry, 03.07.2019 02:00 AquaTyrant9232
Achemist measures the amount of hydrogen gas produced during an experiment. she finds that 926. g of hydrogen gas is produced. calculate the number of moles of hydrogen gas produced.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:30
If a solution is considered basic, then a) the hydroxide ion and hydronium ion concentrations are equal. b) the hydroxide ion concentration is less than the hydronium ion concentration. c) the hydronium ion concentration is greater than the hydroxide ion concentration. d) the hydroxide ion concentration is greater than the hydronium ion concentration.
Answers: 1

Chemistry, 22.06.2019 11:00
What is the molar mass of a gas that has density of 2.054 g/l
Answers: 2

Chemistry, 22.06.2019 16:50
Which element is least likely to undergo a chemical reaction
Answers: 3

Chemistry, 22.06.2019 17:10
Increasing the substrate concentration in an enzymatic reaction could overcome which of the following? a) the need for a coenzymeb) allosteric inhibitionc) competitive inhibitiond) insufficient cofactors
Answers: 1
You know the right answer?
Achemist measures the amount of hydrogen gas produced during an experiment. she finds that 926. g of...
Questions

Business, 25.07.2019 03:00



History, 25.07.2019 03:00


Mathematics, 25.07.2019 03:00




Mathematics, 25.07.2019 03:00

Mathematics, 25.07.2019 03:00


Mathematics, 25.07.2019 03:00


Mathematics, 25.07.2019 03:00



Mathematics, 25.07.2019 03:00


Mathematics, 25.07.2019 03:00