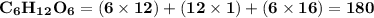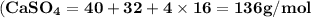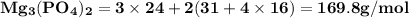Use the periodic table to calculate the molar mass of each compound below. give your answer to the correct number of significant figures. sodium hydroxide (naoh): 2 g/mol water (h2o): 4 g/mol glucose (c6h12o6): 2 g/mol calcium sulfate (caso4): 1 g/mol magnesium phosphate (mg3(po4)2): 1 g/mol

Answers: 2


Another question on Chemistry



Chemistry, 23.06.2019 01:00
Iron (fe) reacts with copper sulfate (cuso4) to form iron (ii) sulfate. in this reaction, cu2+ gains electrons to form cu. which statement is true about this reaction? fe(s) + cuso4(aq) → feso4(aq) + cu(s)
Answers: 3

Chemistry, 23.06.2019 08:20
At which temperature would a reaction with ah= -220 kj/mol and as=-0.05 kj/(mol-k) be spontaneous?
Answers: 2
You know the right answer?
Use the periodic table to calculate the molar mass of each compound below. give your answer to the c...
Questions




English, 25.10.2020 01:50


Chemistry, 25.10.2020 02:00



Business, 25.10.2020 02:00


Mathematics, 25.10.2020 02:00

Medicine, 25.10.2020 02:00

Chemistry, 25.10.2020 02:00

History, 25.10.2020 02:00




Arts, 25.10.2020 02:00

Chemistry, 25.10.2020 02:00

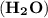 = (2 x 1) + 16 = 18 g/mol
= (2 x 1) + 16 = 18 g/mol