
Chemistry, 24.03.2022 14:00 lollipop83
Determine the reducing agent in the following reaction. 2 K(s) + Cu(C2H302)2(aq) + 2 KC2H2O2(aq) + Cu(s)
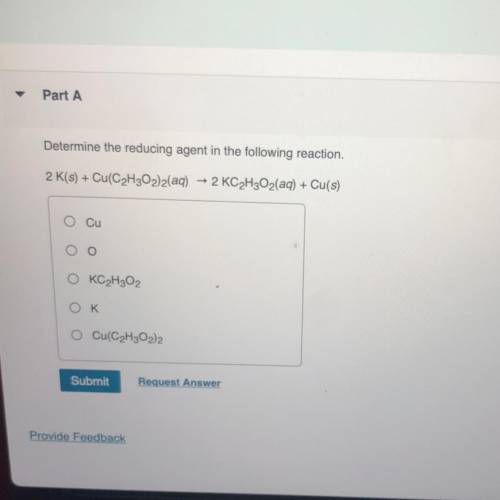

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:00
Agas occupies 475 cm^3 at 313k. find its volume at 367k. you must show all of your work to receive credit. be sure to identify which of the gas laws you will be using
Answers: 2

Chemistry, 22.06.2019 20:00
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3


Chemistry, 23.06.2019 06:20
An object of mass 10.0 kg and volume 1000 ml and density 10 g/ml sinks in water who’s density is 1.0 g/ml. what is the mass of the water which has been displaced in kilograms
Answers: 1
You know the right answer?
Determine the reducing agent in the following reaction.
2 K(s) + Cu(C2H302)2(aq) + 2 KC2H2O2(aq) +...
Questions


English, 07.04.2020 02:55

English, 07.04.2020 02:55


Mathematics, 07.04.2020 02:55








Biology, 07.04.2020 02:55




Biology, 07.04.2020 02:55

Biology, 07.04.2020 02:55

History, 07.04.2020 02:55

History, 07.04.2020 02:55



