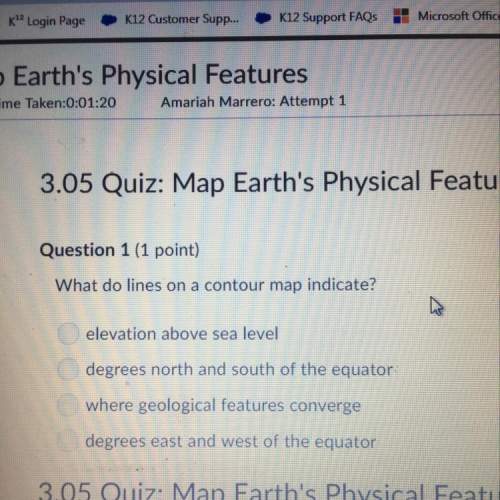
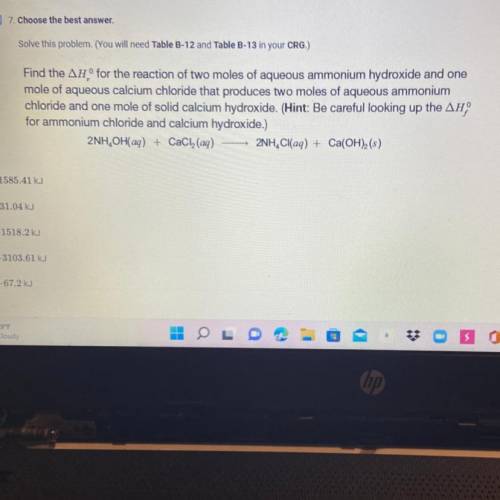
Find the AH° for the reaction of two moles of aqueous ammonium hydroxide and one
mole of aqueous calcium chloride that produces two moles of aqueous ammonium
chloride and one mole of solid calcium hydroxide. (Hint: Be careful looking up the AH
for ammonium chloride and calcium hydroxide.)
2NH, OH(aq) + CaCl(aq) 2NH, Cl(aq) + Ca(OH)(s)
0.-1585.41 kJ
O-31.04 kJ
O-1518.2 kJ
-3103.61 kJ
-67.2 kJ
I NEED HELP FAST !!

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 12:00
Ageochemist examines a piece of metal that he found in the soil. he performs tests to identify the metal from its density, electrical conductivity, and melting point. which statement best describes his investigation? a. he is determining physical properties that are sufficient to identify the metal.b. he is determining chemical properties that are sufficient to identify the metal.c. he is determining physical properties that are insufficient to identify the metal.d. he is determining chemical properties that are insufficient to identify the metal.
Answers: 3

Chemistry, 22.06.2019 12:30
Which statement is true about this reaction? 14n+1h 15o it is a practical source of energy on earth. it occurs only outside the solar system. its product is heavier than each of its reactants. it shows the critical mass of an element.
Answers: 2

You know the right answer?
Find the AH° for the reaction of two moles of aqueous ammonium hydroxide and one
mole of aqueous c...
Questions

Mathematics, 12.03.2022 22:20

Biology, 12.03.2022 22:20




Mathematics, 12.03.2022 22:30

Social Studies, 12.03.2022 22:30

Mathematics, 12.03.2022 22:30


Mathematics, 12.03.2022 22:30




Social Studies, 12.03.2022 22:30

History, 12.03.2022 22:30


English, 12.03.2022 22:30

Biology, 12.03.2022 22:30


Mathematics, 12.03.2022 22:30