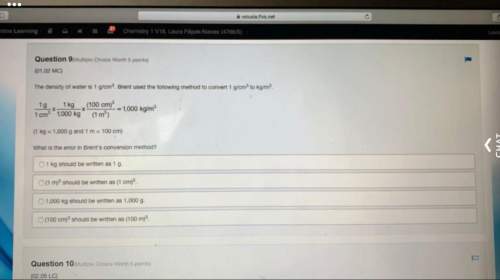
Chemistry, 23.02.2022 05:00 taylormack60
Describing types of chemical bonds Match each bond with the rearrangement of the valance electrons needed to form that bond metallic bond ionic bond covalent bond sharing of electrons sharing of many electrons transferring of electrons
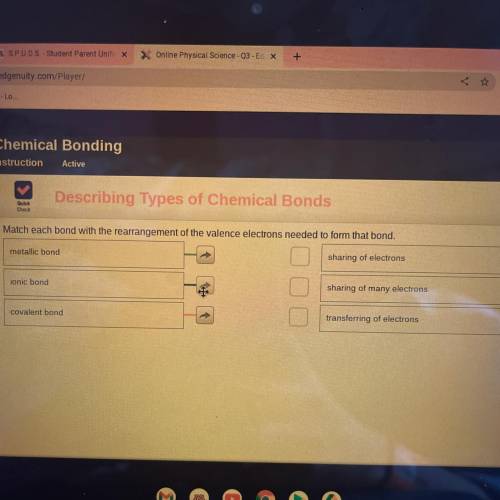

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:30
The table describes how some substances were formed substance 19 description formed by boiling pure water formed by combining three hydrogen atoms to every nitrogen atom formed by adding 5 g of sugar to 1 l of water formed by compressing carbon under high pressure based on the given descriptions, which substance is most likely a mixture?
Answers: 1

Chemistry, 22.06.2019 12:40
In the following table, all the columns for the element calcium are filled out correctly. element electron structure of atom electron structure of ion net ionic charge calcium 1s22s22p63s23p64s2 1s32s22p63s23p64s1 +1 true false
Answers: 2


Chemistry, 22.06.2019 20:00
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1
You know the right answer?
Describing types of chemical bonds Match each bond with the rearrangement of the valance electrons n...
Questions


History, 25.07.2019 15:00




Physics, 25.07.2019 15:00

Biology, 25.07.2019 15:00




Social Studies, 25.07.2019 15:00

Mathematics, 25.07.2019 15:00

Mathematics, 25.07.2019 15:00


Health, 25.07.2019 15:00


Mathematics, 25.07.2019 15:00



History, 25.07.2019 15:00