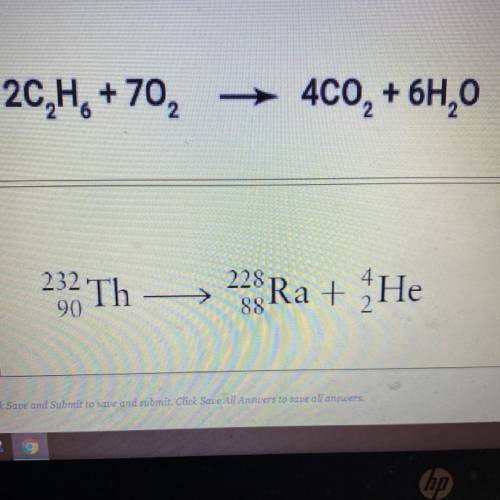Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2


Chemistry, 22.06.2019 14:50
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3

Chemistry, 22.06.2019 20:20
Which formula equation represents the burning of sulfur to produce sulfur dioxide? s(s) + o2(g) 4502(9) 2h2s(s) + 302(g) —> 2h20(0) + 2502(9) 4fes2+1102 —> 2fe2o3 + 8502 2802(g) + o2(9) v205 , 2503(9)
Answers: 1
You know the right answer?
There are two equations shown below. Click on the equation that represents a nuclear reaction?
Questions




Mathematics, 05.01.2021 23:50

Mathematics, 05.01.2021 23:50

Mathematics, 05.01.2021 23:50

Chemistry, 05.01.2021 23:50


Mathematics, 05.01.2021 23:50

Mathematics, 05.01.2021 23:50



World Languages, 05.01.2021 23:50

History, 05.01.2021 23:50


Mathematics, 05.01.2021 23:50

Social Studies, 05.01.2021 23:50

Mathematics, 05.01.2021 23:50

Chemistry, 05.01.2021 23:50