Chemistry, 06.02.2022 05:00 consueloquintan1
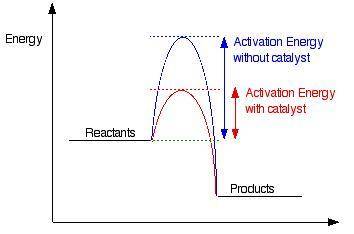
3) Explain the working principle of catalyst for both rate of reaction and equilibrium reaction . show by means of graphical representation

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 00:00
The p sub shell can hold up to 8 electrons in an atom. true or false?
Answers: 1

Chemistry, 22.06.2019 16:30
How many grams of mgbr2 are needed to produce 75g or metal?
Answers: 1

Chemistry, 22.06.2019 21:30
Which of the following changes will decrease the total amount of gaseous solute able to be dissolved in a liter of liquid water? (2 points) decreasing temperature decreasing pressure decreasing surface area decreasing solute concentration
Answers: 1

Chemistry, 23.06.2019 00:30
•hydration •dissociation •dissolving which one goes to which
Answers: 1
You know the right answer?
3) Explain the working principle of catalyst for both rate of reaction and equilibrium reaction . sh...
Questions

History, 23.06.2019 09:00



English, 23.06.2019 09:00



Mathematics, 23.06.2019 09:00

Mathematics, 23.06.2019 09:00


Social Studies, 23.06.2019 09:00




Mathematics, 23.06.2019 09:00

History, 23.06.2019 09:00

Computers and Technology, 23.06.2019 09:00


Mathematics, 23.06.2019 09:00

English, 23.06.2019 09:00