Chemistry, 12.01.2022 14:00 emmaogle2003
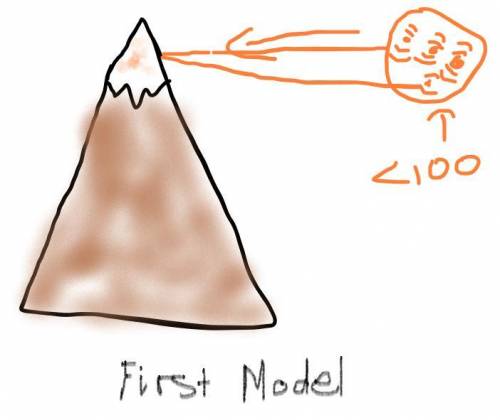
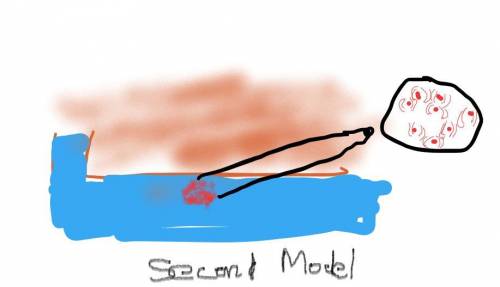
Air pressure decreases as elevation increases. Identify how the boiling point of water on top of a mountain would be different from its boiling point at sea level. Draw two models to show how the particle motion and the state of water on the mountain top and at sea level would change if you kept adding thermal energy to water that was already at 100°C. Label your models to show what happens to the temperature as the energy is added.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:30
This element exists in adundance in the sun.explain how you would go about capturing sunlight.would this captured sunlight contain any of the element?
Answers: 1

Chemistry, 22.06.2019 09:30
Based on its chemical properties, identify the position of each chemical family on the periodic table.
Answers: 3

Chemistry, 22.06.2019 14:00
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3

Chemistry, 22.06.2019 17:10
Increasing the substrate concentration in an enzymatic reaction could overcome which of the following? a) the need for a coenzymeb) allosteric inhibitionc) competitive inhibitiond) insufficient cofactors
Answers: 1
You know the right answer?
Air pressure decreases as elevation increases. Identify how the boiling point of water on top of a m...
Questions


Mathematics, 22.10.2019 23:00

Mathematics, 22.10.2019 23:00

Mathematics, 22.10.2019 23:00


Mathematics, 22.10.2019 23:00


Mathematics, 22.10.2019 23:00






Chemistry, 22.10.2019 23:00





Mathematics, 22.10.2019 23:00

Health, 22.10.2019 23:00