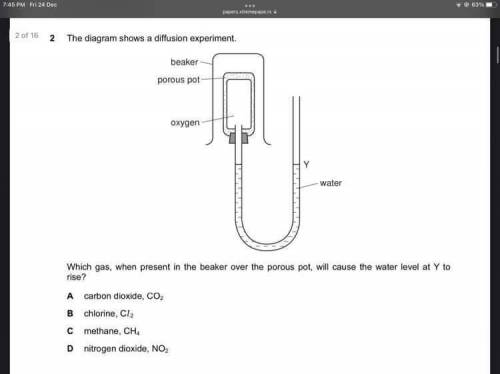
Answer this bs for me pls
...

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 01:30
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3

Chemistry, 22.06.2019 08:00
Define dew point. i am writing this part to be able to ask the question
Answers: 1

Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 3
You know the right answer?
Questions


Mathematics, 27.07.2021 05:10







Mathematics, 27.07.2021 05:10


Social Studies, 27.07.2021 05:10


Computers and Technology, 27.07.2021 05:10

Computers and Technology, 27.07.2021 05:10


Social Studies, 27.07.2021 05:10

Mathematics, 27.07.2021 05:20

Computers and Technology, 27.07.2021 05:20