Help me pls
words from the video:
What do fireworks, a fried egg, and a rusting vehicl...

Chemistry, 13.12.2021 21:40 hectorav6619
Help me pls
words from the video:
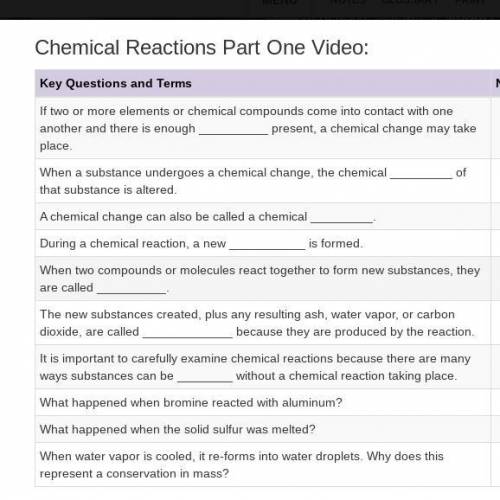
What do fireworks, a fried egg, and a rusting vehicle have in common? They all involve chemical reactions. If two or more elements or chemical compounds come into contact with one another and there is enough energy present, a chemical change may take place.
When a substance undergoes a chemical change, the chemical structure of that substance is altered. A chemical change can also be called a chemical reaction. During a chemical reaction a new substance is formed.
Observe the chemical reaction between bromine and aluminum. Pieces of aluminum are cut from a strip of foil and added to liquid bromine. At first there is no reaction because of the protective oxide coating on the aluminum. But soon an exothermic reaction between bromine and aluminum causes brown fumes of bromine to be given off. But as a new substance, aluminum bromide is produced. White smoke is produced.
The resulting residue is also aluminum bromide. When two compounds or molecules react together to form new substances, they are called reactants. They new substances created plus any resulting ash, water vapor, or carbon dioxide are called products, because they are produced by the reaction.
Some chemical reactions, such as the explosion of fireworks, can happen very fast—in less than a second.
Others, like the rusting of a car, might take years. It is important to carefully examine chemical reactions because there are many ways substances can be changed without a chemical reaction taking place.
Changes of state, for example, from a solid to a liquid or from a liquid to a gas, always involve a physical change rather than a chemical one. Melting sulfur is a good example. When solid sulfur is heated to 120 degrees Celsius it melts and first forms a pale yellow liquid.
In this form the atoms are covalently bonded and the liquid can flow easily. However, heated up beyond 150 degrees Celsius, the result is a dark red liquid that is over 2,000 times stickier. The sulfur may have changed physically but it's still sulfur.
No chemical reaction has taken place. Most of the time changes of state can be reversed by either adding or taking away heat. When water is boiled, for example, water vapor molecules are released in the process.
If you captured all the vapor molecules in a container they would reform again as water droplets once the vapor cooled. When a chemical change occurs it is more difficult to revert the substance back to its original state.


Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:00
Oxidation-reduction reactions (often called "redox" for short) are reactions that involve the transfer of electrons from one species to another. oxidation states, or oxidation numbers, allow chemists to keep track of these electron transfers. in general, one element will lose electrons (oxidation), with the result that it will increase in oxidation number, and another element will gain electrons (reduction), thereby decreasing in oxidation number. the species that is oxidized is called the reducing agent or reductant. the species that is reduced is called the oxidizing agent or oxidant. to sum up: oxidation = increase in oxidation state = loss of electrons = reducing agent reduction = decrease in oxidation state = gain of electrons = oxidizing agent part a which element is oxidized in this reaction? fe2o3+3co→2fe+3co2 enter the elemental symbol. view available hint(s) is oxidized part b which element is reduced in this reaction? 2hcl+2kmno4+3h2c2o4→6co2+2mno2+2kcl+4h2o enter the elemental symbol. view available hint(s) is reduced
Answers: 1

Chemistry, 22.06.2019 17:40
Which statement about hf is true? it is zero for any compound in its standard state. it is positive when the bonds of the product store more energy than those of the reactants. it is negative when a compound forms from elements in their standard states. it is zero for any element that is in the liquid state.
Answers: 1

Chemistry, 22.06.2019 18:10
Consider the following reaction at equilibrium: c(s)+h2o(g)⇌co(g)+h2(g) predict whether the reaction will shift left, shift right, or remain unchanged upon each of the following disturbances. a) c is added to the reaction mixture. b) h2ois condensed and removed from the reaction mixture c) co is added to the reaction mixture d) h2 is removed from the reaction mixture.
Answers: 3

Chemistry, 22.06.2019 20:00
Suppose that some of the compound spilled out of the crucible after it was heated. would that cause the percent by mass of water in the compound determined by the experiment to be too low, too high, or unchanged? briefly explain your answer.
Answers: 1
You know the right answer?
Questions

History, 24.08.2019 11:00


Mathematics, 24.08.2019 11:00



Mathematics, 24.08.2019 11:00





Biology, 24.08.2019 11:00



Biology, 24.08.2019 11:00

World Languages, 24.08.2019 11:00




Mathematics, 24.08.2019 11:00

Biology, 24.08.2019 11:00



