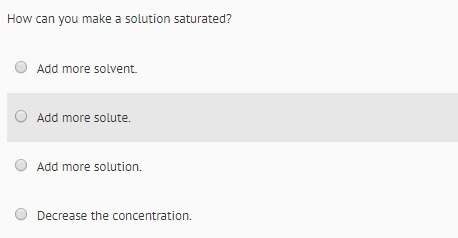
Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:00
Asample of aluminum foil contains 8.60 × 1023 atoms. what is the mass of the foil?
Answers: 1

Chemistry, 22.06.2019 06:00
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1

Chemistry, 23.06.2019 12:30
You have 125 g of a certain seasoning and are told that it contains 70.0 g of salt. what is the peroentage of salt by mass in this seasoning?
Answers: 2

Chemistry, 24.06.2019 00:30
Which will not appear in the equilibrium constant expression for the reaction below
Answers: 3
You know the right answer?
Determine how many moles of gas are in the flask with slope of temperature and pressure....
Questions

Advanced Placement (AP), 20.01.2021 14:00



Physics, 20.01.2021 14:00

Mathematics, 20.01.2021 14:00





Mathematics, 20.01.2021 14:00


Chemistry, 20.01.2021 14:00




Mathematics, 20.01.2021 14:00

Mathematics, 20.01.2021 14:00

English, 20.01.2021 14:00

History, 20.01.2021 14:00

Mathematics, 20.01.2021 14:00